|
|
|
|

| Backgrounders and Facts | |
| More Publications | |
| Search our Database | |
| Industry Data |
![]()
 |
On the threshold of the new millennium, researchers press their search for cures for cancer, Alzheimer's, heart disease, stroke, and many other diseases. But we will find these cures only if the public and private sectors continue to provide strong support for biomedical research.
To this end, PhRMA advocates increased funding for the basic research conducted by the National Institutes of Health and strives to ensure a favorable climate for private-sector research.
In the past year, PhRMA has helped to improve the climate for biomedical research in several ways. Working with other high-technology industries, for example, PhRMA helped to gain an extension of the R&D tax credit. Currently, PhRMA is cooperating in efforts to make the credit permanent.
A study conducted by Coopers and Lybrand showed that a permanent R&D tax credit would add about $4 billion to the amount the pharmaceutical industry would otherwise spend on R&D over the next 12 years. This increased research would accelerate the discovery and development of breakthrough new medicines that save lives and money, by reducing the need for alternative, more expensive treatments such as hospitalization and nursing home care.
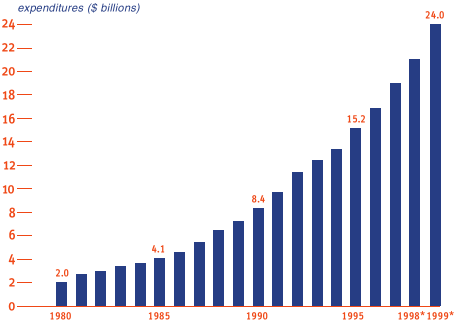
R&D Investments by Research-Based Pharmaceutical Companies |
| *estimated Source: PhRMA Annual Survey, 1999 Every five years since 1980, U.S. pharmaceutical companies have practically doubled spending on R&D. In 1999, companies expect to invest $2 billion to discover and develop new medicines - a 14.1 percent increase over 1998's record-setting $21.1 billion. Research-based pharmaceutical companies pour back $1 out of every $5 in domestic sales into R&D - a higher percentage than any other industry. |
Widespread public concern about the confidentiality of patient records has led to the development of legislative proposals that could slow the discovery and development of new cures and treatments. On the state, federal and international levels, proposals were made that could seriously undermine pharmaceutical innovation by restricting the access of scientists to patient data.
More than 50 bills dealing with patient confidentiality were introduced in 23 states. Stressing the importance of biomedical research, the critical need of researchers for access to medical information, and the care with which pharmaceutical companies protect the confidentiality of patient-identifiable information, PhRMA helped to defeat 23 of these bills and continues to work to block or modify many others. At the federal level, Congress, striving to act before a legislatively-imposed deadline, has considered five bills on medical privacy, some of which would make it difficult or impossible to conduct cutting-edge research. PhRMA has helped to mitigate the negative impact of proposed legislation on medical research, pointing out that, while patients deserve to have their medical records kept private, they also deserve to have their unmet medical needs met.
Internationally, PhRMA is working with the European Federation of Pharmaceutical Industry Associations (EFPIA) to achieve a broad exemption from the European Union Privacy Directive in order to protect pharmaceutical research.
| to the next page of PhRMA Annual Report 1999-2000 | |
| to the first page of PhRMA Annual Report 1999-2000 |
Privacy
Policy • Members
Site • Contact
Us • Site
Credits • Site Map • Employment
Pharmaceutical Research and Manufacturers of America
1100 Fifteenth
Street, NW Washington, DC 20005
+1.202.835.3400